Scientists achieved new progress in Micro/Nanostructured Ceria and Novel Bifunctional Catalysts for Lithium-air Batteries
Date:26-10-2012 Print
Cerium oxide (CeO2) is a technologically important material due to its wide applications as a promoter in three-way catalysts (TWCs) for the elimination of toxic auto-exhaust gases, low-temperature water-gas shift (WGS) reaction, fuel cells, oxygen sensors, oxygen permeation membrane systems, ultraviolet absorbent, as well as biotechnology, environmental chemistry, and medicine. In particular, nanostructured ceria-based materials show some unique properties compared to the coarsened bulk materials, including increased electronic conductivity, the size-induced lattice relaxation, the pressure-induced phase transformation, and the blue shift in ultraviolet absorption spectra, etc.
In recent years, Dr. SUN Chunwen and his colleagues at Beijing National Laboratory for Condensed Matter Physics, Institute of Physics, Chinese Academy of Sciences have made progress in the synthesis of CeO2 materials, including one dimensional nanowires and nanorods (Nanotechnology, 16, 1454 (2005); Chem. Lett. 662 (2004)) as well as flowerlike mesoporous microspheres with micro/nanostructure (J. Phys. Chem. B, 110, 13445 (2006);Micropor. Mesopor. Mater. 120, 426 (2009). They also studied the applications of these materials in solid oxide fuel cells and CO oxidation (Electrochem. Commun, 8, 833 (2006); J. Phys. Chem. Solid, 68, 1785 (2007)).
Recently, they were invited by the editor of Energy & Environmental Science to write a review paper on nanostructured ceria-based materials. In this critical review, the recent progress in the preparation, properties, new characterization approaches, and theoretical studies of nanostructured ceria-based materials has been highlighted. Some typical applications regarding ceria-based nanomaterials have also been demonstrated (Figure 1). Remarks on the challenges and perspectives on this exciting field have been proposed as well. This paper was published in Energy & Environmental Science 5, 8475 (2012).
At present, the ever-increasing demand for energy has stimulated the developments of low-cost, efficient, and environmentally benign energy storage and conversion devices. Oxygen-reduction reaction (ORR) and oxygen-evolution reaction (OER) are the core processes for renewable energy technologies, such as, fuel cells, lithium-air batteries and water splitting for hydrogen production. For Li/air batteries with an organic electrolyte, a theoretical specific energy density of 11140 Wh kg-1 can be reached if oxygen is accessed directly from the air, much higher than those of state-of-the-art lithium-ion batteries and other energy storage devices. However, the discharge product Li2O2 can gradually clogs the porous electrode to degrade the performance. A lithium-air system with hybrid electrolytes (Figure 2), which units an O2 reduction electrode in aqueous electrolyte and a lithium anode in organic electrolyte by a lithium super-ionic conductor ceramic film (LISICON), can solve this problem. To make Li-air batteries practical for commercial applications, many issues still need to be addressed, including low electrolyte stability, poor round-trip efficiency, rate capability, and cycle life.
Due to its high electronic conductivity, large surface area and appropriate pore structure, carbon black (commercial XC-72R and Ketjen carbon) is the commonly used support for ORR catalysts in Li-air batteries and proton-exchange-membrane (PEM) fuel cells. However, the carbon support of the costly noble-metal catalysts is thermodynamically unstable above 0.207 V versus the normal hydrogen electrode (NHE). Oxidation of a carbon support, known as carbon corrosion, has been studied in PEM fuel cells; it decreases performance by accelerating the loss of active surface area, altering pore morphology and surface characteristics, either dropping noble-metal nanoparticles from the carbon-support surface or causing their aggregation into large particles, and changing the surface hydrophobicity which can cause gas-transport difficulties. However, this issue has not yet attracted attention in the field of Li-air batteries. Dr. SUN Chunwen and Prof. CHEN Liquan at IOP, collaborated with Prof. John B. Goodenough at UT Austin and Prof. Youngsik Kim at Indiana University Purdue University, developed an low-cost, efficient and stable bifunctional catalysts Sr0.95Ce0.05CoO3-δ (SCCO)-Cu nanocomposite for the ORR and OER in aqueous solution. The performance of the SCCO-based catalysts in Li-air batteries is better at higher current rates (?0.1 mA cm-2) than that of Vulcan XC-72 and even close to that of the 50% Pt/carbon-black catalyst. The improved performance of the SCCO-Cu catalyst can be ascribed to the synergetic effect of the SCCO and copper. Those results have been published in Journal of Materials Chemistry 22, 18902 (2012)。
These works above are financially supported by the National Science Foundation of China (NSFC) (Grant No. 51172275), the National Key Basic Research Program of China (Grant No. 2012CB215402), and the Institute of Physics start-up funding for the talents.
In recent years, Dr. SUN Chunwen and his colleagues at Beijing National Laboratory for Condensed Matter Physics, Institute of Physics, Chinese Academy of Sciences have made progress in the synthesis of CeO2 materials, including one dimensional nanowires and nanorods (Nanotechnology, 16, 1454 (2005); Chem. Lett. 662 (2004)) as well as flowerlike mesoporous microspheres with micro/nanostructure (J. Phys. Chem. B, 110, 13445 (2006);Micropor. Mesopor. Mater. 120, 426 (2009). They also studied the applications of these materials in solid oxide fuel cells and CO oxidation (Electrochem. Commun, 8, 833 (2006); J. Phys. Chem. Solid, 68, 1785 (2007)).
Recently, they were invited by the editor of Energy & Environmental Science to write a review paper on nanostructured ceria-based materials. In this critical review, the recent progress in the preparation, properties, new characterization approaches, and theoretical studies of nanostructured ceria-based materials has been highlighted. Some typical applications regarding ceria-based nanomaterials have also been demonstrated (Figure 1). Remarks on the challenges and perspectives on this exciting field have been proposed as well. This paper was published in Energy & Environmental Science 5, 8475 (2012).
At present, the ever-increasing demand for energy has stimulated the developments of low-cost, efficient, and environmentally benign energy storage and conversion devices. Oxygen-reduction reaction (ORR) and oxygen-evolution reaction (OER) are the core processes for renewable energy technologies, such as, fuel cells, lithium-air batteries and water splitting for hydrogen production. For Li/air batteries with an organic electrolyte, a theoretical specific energy density of 11140 Wh kg-1 can be reached if oxygen is accessed directly from the air, much higher than those of state-of-the-art lithium-ion batteries and other energy storage devices. However, the discharge product Li2O2 can gradually clogs the porous electrode to degrade the performance. A lithium-air system with hybrid electrolytes (Figure 2), which units an O2 reduction electrode in aqueous electrolyte and a lithium anode in organic electrolyte by a lithium super-ionic conductor ceramic film (LISICON), can solve this problem. To make Li-air batteries practical for commercial applications, many issues still need to be addressed, including low electrolyte stability, poor round-trip efficiency, rate capability, and cycle life.
Due to its high electronic conductivity, large surface area and appropriate pore structure, carbon black (commercial XC-72R and Ketjen carbon) is the commonly used support for ORR catalysts in Li-air batteries and proton-exchange-membrane (PEM) fuel cells. However, the carbon support of the costly noble-metal catalysts is thermodynamically unstable above 0.207 V versus the normal hydrogen electrode (NHE). Oxidation of a carbon support, known as carbon corrosion, has been studied in PEM fuel cells; it decreases performance by accelerating the loss of active surface area, altering pore morphology and surface characteristics, either dropping noble-metal nanoparticles from the carbon-support surface or causing their aggregation into large particles, and changing the surface hydrophobicity which can cause gas-transport difficulties. However, this issue has not yet attracted attention in the field of Li-air batteries. Dr. SUN Chunwen and Prof. CHEN Liquan at IOP, collaborated with Prof. John B. Goodenough at UT Austin and Prof. Youngsik Kim at Indiana University Purdue University, developed an low-cost, efficient and stable bifunctional catalysts Sr0.95Ce0.05CoO3-δ (SCCO)-Cu nanocomposite for the ORR and OER in aqueous solution. The performance of the SCCO-based catalysts in Li-air batteries is better at higher current rates (?0.1 mA cm-2) than that of Vulcan XC-72 and even close to that of the 50% Pt/carbon-black catalyst. The improved performance of the SCCO-Cu catalyst can be ascribed to the synergetic effect of the SCCO and copper. Those results have been published in Journal of Materials Chemistry 22, 18902 (2012)。
These works above are financially supported by the National Science Foundation of China (NSFC) (Grant No. 51172275), the National Key Basic Research Program of China (Grant No. 2012CB215402), and the Institute of Physics start-up funding for the talents.
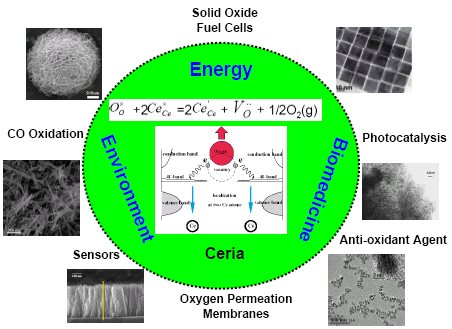 |
| Figure 1. Nanostructured Ceria materials and their applications(Image by SUN Chunwen et al) |
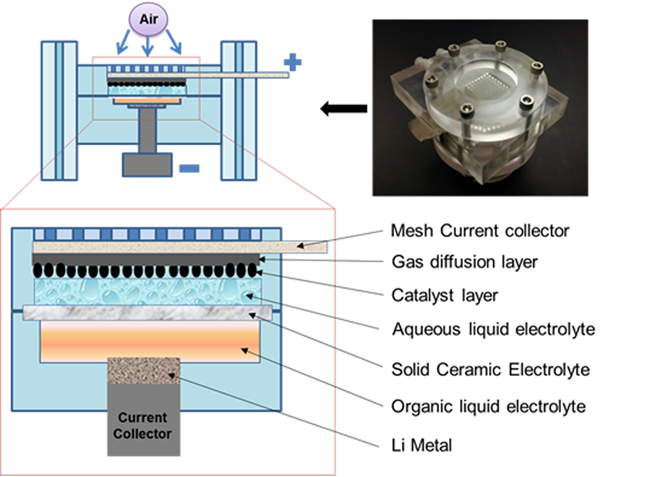 |
| Figure 2. Schematic diagram of a laboratory-sized aqueous Li-air battery with hybrid electrolytes(Image by SUN Chunwen et al) |
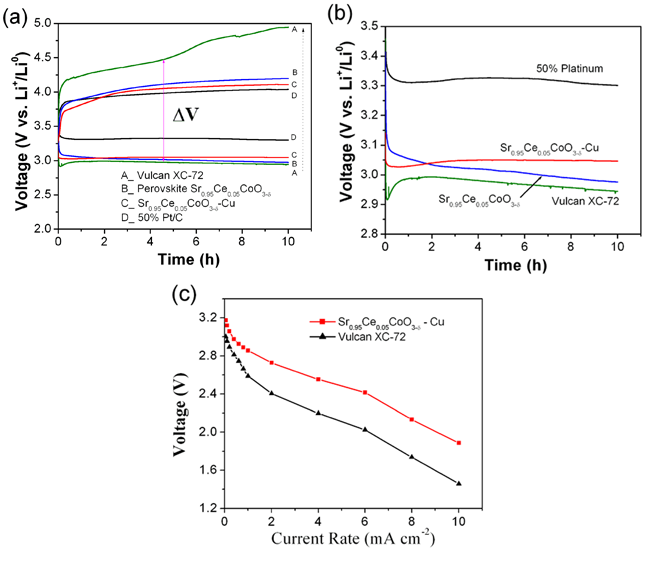 |
| Figure 3. (a)The first discharge-charge curves of the prepared lithium-air batteries with different catalysts, (b) magnified discharge curves to clearly show the difference among them in (a), and (c) comparison of discharge voltage as a function of current rates between Vulcan XC-72 and Sr0.95Ce0.05CoO3-δ-Cu catalysts. (Image by SUN Chunwen et al) |
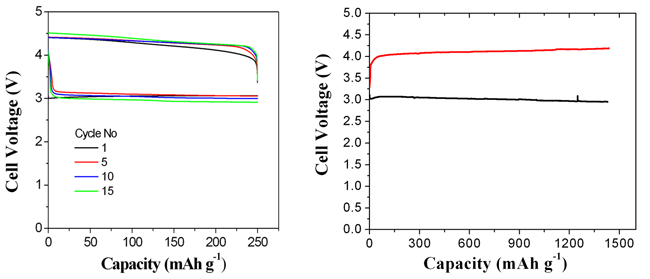 |
| Figure 4. (left)The discharge-charge curves of the prepared lithium-air batteries with the SCCO-Cu catalyst at a current density of 0.2 mA/cm2at different cycles, (right) Voltage versus discharge-charge capacity for the lithium-air batteries with the SCCO-Cu catalyst(Image by SUN Chunwen et al) |


