Research Breakthroughs Provide Microscopic Insights into Surface Wetting
Date:28-04-2013 Print
The wetting property of water has been a topic of wide interest due to its central role in numerous processes in physical, biological, chemical, and technological systems, including artificial rainfall and protein folding. Understanding the relevance between the wetting properties and the structure of interface is a key to study surface wetting. In 1947, Vonnegut proposed to use small particles of silver iodide AgI, based on lattice match between AgI and ice, to promote water nucleation in clouds in forming precursors for artificial rainfall. However, subsequent experimental studies seem to show that there is no obvious influence of surface lattice constant on ice formation. To this day, the precise causal relation between surface lattice and water wetting remains ambiguous.
Prof. MENG Sheng and coworkers at the Beijing National Laboratory for Condensed Matter Physics and Institute of Physics, Chinese Academy of Sciences have now studied systematically the effect of surface lattice on water contact angles based on molecular dynamics (MD) simulations of water droplets on model fcc (111) crystal surface. They reported that a small uniform strain (±3%) applied to the lattice constant of a multilayer hydrophilic surface can introduce a marked change in the wetting tendency, in a paper recently published in Physical Review Letters [Phys. Rev. Lett. 110, 126101 (2013)].
When the lattice constant of a hydrophilic surface matches the projected oxygen-oxygen distance of bulk water to the surface, a contact-angle minimum is resulted in their simulations.They also found evidences for this peculiar behavior backed up by some early experimental data. In stark contrast, such a lattice strain has little effect on the wetting properties of hydrophobic surfaces, whose hydrophobicity continues to increase with the increasing tensile strain as a result of declined surface energy.
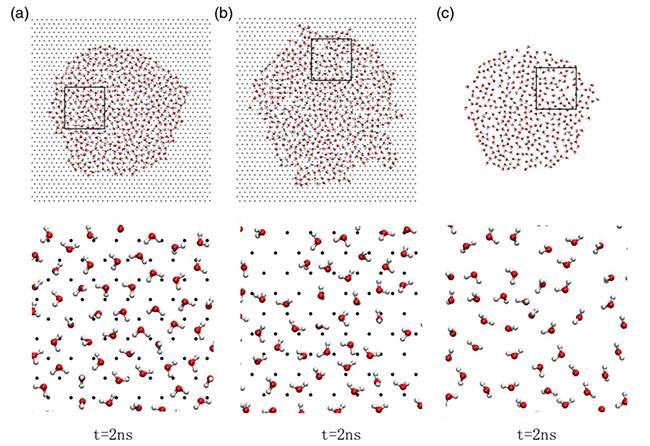
“This is because of the structure of the first water layer next to the hydrophilic surface gradually loses characteristics of liquid water when moving away from the contact-angle minimum (see Fig. 1 and Fig. 2)”, explains MENG Sheng, the leading author of this work, “in contrast, interactions between water and atoms of a hydrophobic surface are relatively weak, and the structure of interfacial water is little disturbed”. This work demonstrates a close correlation among the length of lattice constant, contact angle of the water droplet, and the structure and dynamics of vicinal water, and provides clues towards tuning hydrophilic properties of solid surfaces mechanically.
The work was partly supported by the water-projects cluster, and NSFC (Grants No. 11074287 and No. 11222431) and MOST (2012CB921403).
CONTACT:
Prof. MENG Sheng
Institute of Physics, Chinese Academy of Sciences
Email: smeng@ iphy.ac.cn Tel:86-10-82649396
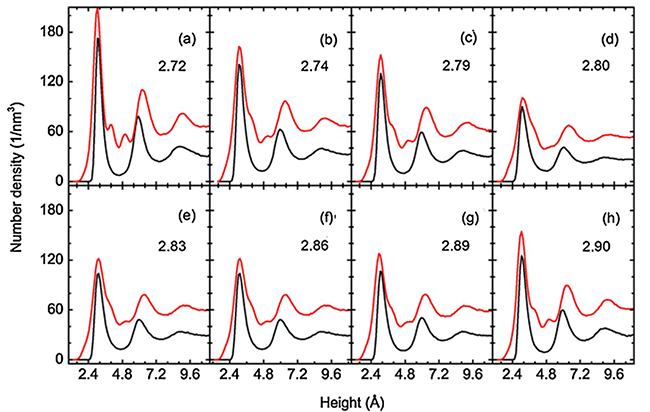 |
| Fig 1 Number density of oxygen (black lines) and hydrogen atoms (red lines) in water droplets near the hydrophilic surface. (Image by Prof. MENG Sheng et al.) |
 |
|
Fig 2 Snapshots of the first water layer on the hydrophilic surface at the end of 2 ns on the surface with
|


