Universal Scaling of Intrinsic Resistivity in Two-Dimensional Metallic Borophene
Date:30-03-2018 Print
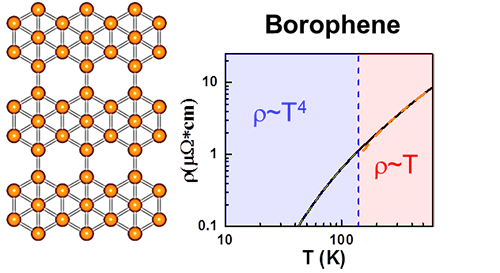
Description: Intrinsic resistivity of borophene is highly dependent on the polymorphs and carrier densities. It is observed that the resistivity is well described using the Bloch-Grüneisen model, exhibiting a universal scaling behavior.
The resistivity of metals originating from electron-phonon (e-ph) interactions (i.e. their intrinsic resistivity ρe-ph) is an important fundamental quantity in condensed matter physics and materials science. Recently, several borophene phases have been synthesized on Ag surfaces, e.g., β12, χ3 and triangle sheets (Nat. Chem. 2016, 8 (6), 563-568; Science 2015, 350, 1513). All experimentally realized borophenes exhibit intrinsic metallic properties, providing an ideal platform to explore the transport properties of two-dimensional (2D) metals. As a unique 2D elemental metal, the intrinsic electrical resistivity of borophene lies at the heart of its potential application in electronic devices and other boron-based nanodevices in the future. Experimental or theoretical investigations on the intrinsic electronic transport properties of borophene as a prototype 2D metal are still lacking.
Now a group from Institute of Physics, Chinese academy of sciences led by Prof. Sheng Meng demonstrates the intrinsic electric resistivity of new-emerging 2D metal----borophene by electron-phonon coupling calculations based on first-principles. They find that the resistivity of borophene is highly dependent on different polymorphs and electron densities of borophenes. Interestingly, it is observed that a universal behavior of the intrinsic resistivity is well described using the Bloch-Grüneisen model. In contrast to graphene and conventional metals, the intrinsic resistivity of borophenes can be easily tuned by adjusting carrier densities, while the Bloch-Grüneisen temperature is nearly pinned at 100 K. This work suggests that monolayer boron can serve as intriguing platform for realizing tunable two-dimensional electronic devices.
Reference:
Zhang et al. Angew. Chem. Int. Ed. 2018, DOI: 10.1002/anie.201800087


