Novel technique enables in situ monitoring isotopic impurities in liquid helium
Date:02-06-2021 Print
Helium is the epitome of quantum mechanics because of its small mass and inertness to react. Its large atomic motions even at absolute zero is guaranteed by the uncertainty principle, leading to a series of exotic quantum phenomena. Helium does not freeze even till absolute zero – unless higher pressure is applied. This unique feature makes liquid and solid helium the purest substances as all other impurities would be frozen out and absorbed by interfaces, with the only exception being its own isotopic impurities, namely 3He in 4He or vice versa. The isotopic impurities are extremely mobile and feature with different quantum statistics, therefore even a tiny amount can significantly change properties of liquid or solid helium. Furthermore, their distribution and behavior are highly sensitive to environment, making their dynamics hard to predict. This is one of the main reasons that quantitative studies on their effects on helium properties are still lacking.
Recently, a group of researchers, led by Prof. Zhi Gang Cheng of Institute of Physics, Chinese Academy of Sciences (IOP-CAS) in collaboration with Prof. John Beamish of University of Alberta, propose a novel and convenient technique for in-situ measurement of isotopic impurity concentration in liquid helium. Even with almost the same polarizability, atomic volumes of the two isotopes of helium are different by about 30%. Based on Clausius-Mossotti relation, this leads to a distinct contrast in their dielectric constants, hence dielectric constant of a 3He/4He mixture can be used as a measure of its impurity concentration. The researchers immersed a concentric capacitance in liquid helium; its dielectric constant can be measured in real-time via capacitance. Liquid pressure was simultaneously measured in order to exclude the capacitance change contributed by pressure variations. Without sophisticated instrument, the resolution can easily reach 40 ppm realized by only using a commercial capacitance bridge. Resolution can be improved by one or two orders of magnitude if more careful homemade bridge is employed.
To test the technique, the researchers prepared solid-liquid coexisting 4He samples, and systematically doped 3He impurities. Using the technique, they observed the aggregation of 3He atoms in the liquid phase at low temperatures, and they migrated to solid phase once temperature was raised. With the benefit of real-time measurement, the dynamics of the migration could be quantitatively studied: the diffusion time constant decreased as temperature was increased, and the behavior can be explained by a thermally activated process; the extracted activation energy suggests that 3He atoms diffuse along grain boundaries in the solid phase.
This novel technique advances the state-of-the-art in monitoring isotopic impurities in liquid helium with unprecedented resolution. With simple design and setup, the technique can be conveniently generalized to a broad range of experimental studies. It enables quantitative studies on dynamics of isotopic impurities and their impact on helium properties.
This study was reported by Physical Review Research entitled “In situ monitoring distribution and migration of 3He in liquid-solid 4He mixtures” (Phys. Rev. Research 3, 023136 (2021)).
The study was supported by Key Research Program of Frontier Sciences, CAS, National Key R&D Program of China, National Science Foundation of China (NSFC), and a grant from NSERC Canada.
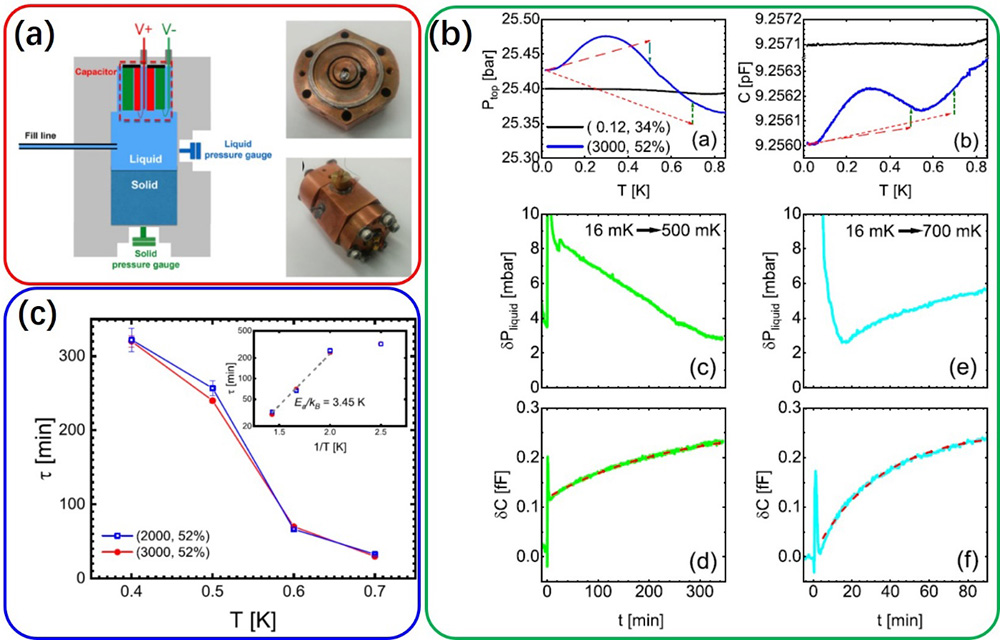
Figure 1 (a) Experimental setup and photos of the experimental cell. (b) Evolutions of liquid pressure and capacitance after temperatures were abruptly raised from 16 mK to higher temperatures (500 and 700 mK, respectively). Capacitance evolutions can be fitted by \(\delta C(t)=\delta C_{0}\left(1-e^{-\left(t-t_{0}\right) / \tau}\right)\), with which the diffusion time constant τ can be extracted. (c) Temperature dependence of diffusion time constants. The inset is the Arrhenius plot of τ vs. 1/T, and the activation energy can be extracted to be 3.45 K.
Contact:
Institute of Physics, Chinese Academy of Sciences
CHENG Zhigang
Email: zgcheng@iphy.ac.cn
Key word:
Helium; Isotopic impurity; dielectric constant
Abstract:
Helium is ideal for studies of quantum liquids and solids, with its significant advantage of being extremely pure at low temperatures—all except isotopic impurities are frozen out. However, even tiny concentrations of isotopic impurities have significant effects on both dynamic and thermodynamic properties. Difficulties in predicting and measuring impurities’ distributions and motions make it hard to systematically study such effects. Here we report a sensitive technique combining dielectric and pressure measurements to resolve concentration changes of isotopic impurities in liquid helium with a resolution of 40 ppm. Using this technique, we observed that 3He impurities concentrate in liquid phase of solid-liquid coexisting 4He at low temperatures, and their migration between the liquid and solid phases when temperature is changed. This migration process is much slower than 3He diffusion in the liquid phase, suggesting the bottleneck is in the solid phase and possibly transmission across the liquid-solid interface.


