Scientists Reveal "Magic Boron Clusters" on Monolayer Borophene
Date:07-03-2023 Print
Boron, which is located adjacent to carbon in the periodic table, has one less electron than carbon. This electron deficiency results in its strong bonding ability and diverse boron polymorphs. Despite their variations, all boron polymorphs have B12 clusters as their building blocks, which possess an icosahedral cage structure. Nonetheless, research combining experimental and theoretical approaches has revealed that small boron clusters in the gas phase, such as Bn (n = 3 to 12, 36), are planar or quasi-planar. This feature has enabled the possibility of two-dimensional (2D) boron, also known as borophene. The unique properties of borophene, such as its mechanical flexibility, metallic Dirac fermions, and superconductivity, have generated significant experimental and theoretical interest.
The planar structure motifs of boron clusters benefit from the strong two-center-two-electron (2c–2e) σ bonds and multicenter-two-electron (nc–2e) bonds, which results in electron delocalization that enhances their chemical stability. Since the formation and arrangement of small boron clusters on a surface represent the initial stage of borophene growth, these clusters can serve as fundamental building blocks and play a crucial role in the formation of monolayer and bilayer borophenes. Moreover, the polymorphic nature of borophene suggests that the growth of boron clusters on metal surfaces can strongly influence the structure of borophene. However, previous experimental studies have mainly focused on small boron clusters in the gas phase, and only a few theoretical studies have investigated the evolution of boron clusters from small clusters to 2D sheets on surfaces. Recently, researchers from the Institute of Physics, Chinese Academy of Sciences, and the University of Science and Technology of China, including Chen Caiyun in Prof. CHEN Lan and Prof. WU Kehui’s groups and LV Haifeng in Prof. WU Xiaojun's group, have revealed the formation of boron clusters with magic numbers on monolayer borophene and observed the evolution process from monolayer borophene with adsorbed boron clusters into bilayer borophene.
Through their research, the scientists discovered that by evaporating boron on a Cu(111) surface covered with a monolayer of borophene, they could create boron clusters with a unique size distribution. Each boron cluster selectively binds to specific sites in each unit cell of the monolayer borophene, resulting in periodic arrangements. Density functional theory (DFT) calculations identified the chemically adsorbed boron clusters as B5 clusters, and the formation and arrangement of these clusters on the surface were found to be the result of the in-plane charge distribution and electron delocalization in the monolayer borophene. Additionally, the close-packed adsorption of B5 clusters on the monolayer borophene was observed to result in the spontaneous transformation into bilayer borophene.
This study entitled "Selective binding and periodic arrangement of magic boron clusters on monolayer borophene" was published on Proceedings of the National Academy of Sciences (PNAS).
The study was supported by the National Science Foundation of China, the Ministry of Science and Technology of China, the Chinese Academy of Sciences and Beijing Natural Science Foundation.
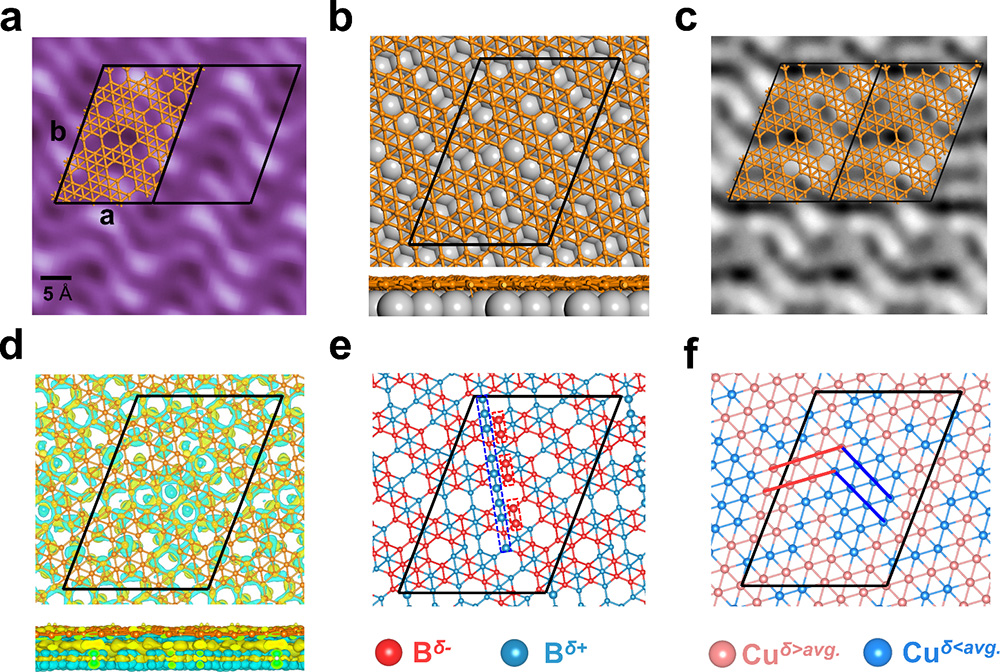
Fig.1 The STM characterization, structural motifs, simulated STM image and charge redistribution of monolayer borophene on Cu(111). (Image by Institute of Physics)
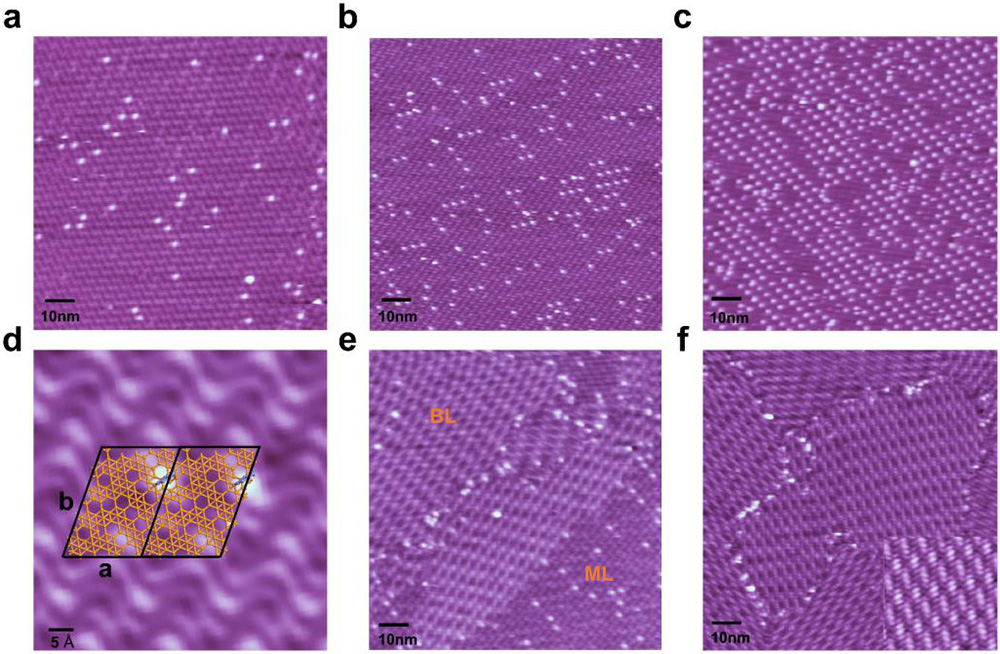
Fig.2 The STM characterization of boron clusters on monolayer borophene and their evolution to bilayer borophene. (Image by Institute of Physics)
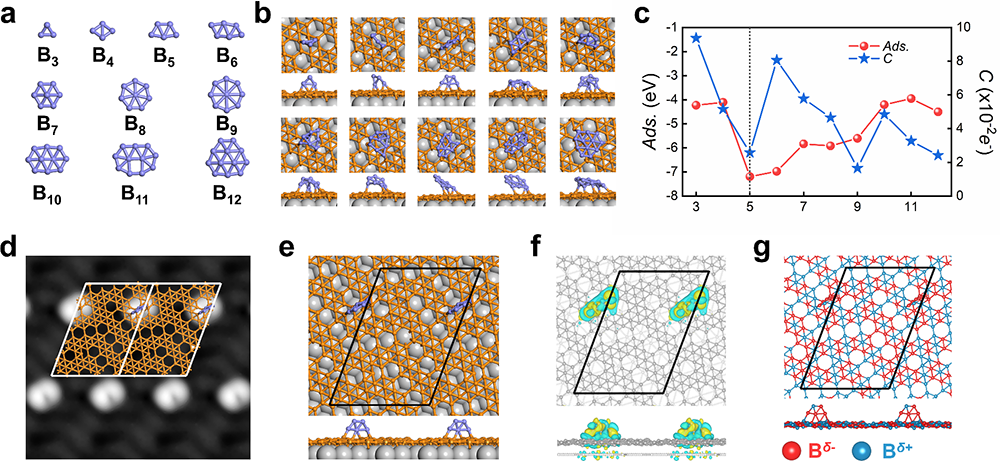
Fig.3 The structures of boron clusters (Bn), calculated structural motifs of monolayer borophene with adsorbed Bn, simulated STM image and the charge redistribution. (Image by Institute of Physics)
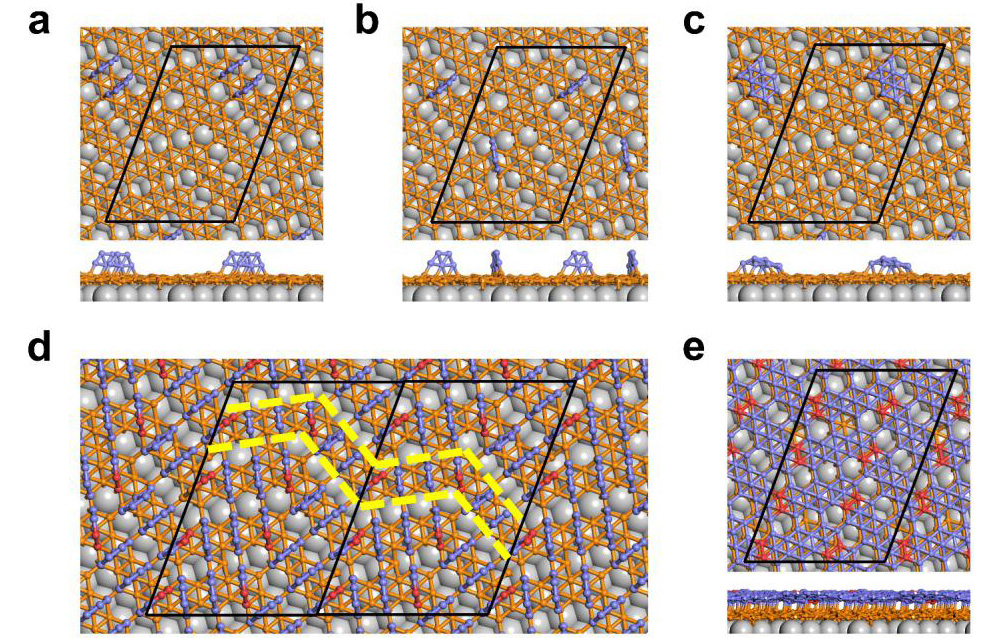
Fig.4 The close-packed adsorption of B5 clusters on monolayer borophene and their transformation to bilayer borophene. (Image by Institute of Physics)
Contact:
Institute of Physics
CHEN Lan
Email:lchen@iphy.ac.cn
Key words:
Boron clusters; scanning tunneling microscopy; molecular beam epitaxy; bilayer borophene; first-principles calculation
Abstract:
The synthesis and characterization of small boron clusters with unique size and regular arrangement are crucial to boron chemistry and the development of two-dimensional borophene materials. In this study, a combination of theoretical calculations and joint molecular beam epitaxy and scanning tunneling microscopy experiments led to the formation of unique B5 clusters on monolayer borophene (MLB) on a Cu(111) surface. The B5 clusters selectively bound to specific sites of the MLB with covalent boron-boron bonds in a periodic arrangement, which was attributed to the charge distribution and electron delocalization character of the MLB and prevented the nearby co-adsorption of B5 clusters. Moreover, the close-packed adsorption of B5 clusters facilitated the synthesis of bilayer borophene, exhibiting a domino effect-like growth mode. The successful growth and characterization of uniform boron clusters on a surface not only enriches the field of boron-based nanomaterials but also sheds light on the crucial role that small clusters play during the growth of borophene.


