Sodium-ion Battery Achieves No Thermal Runaway
Date:09-04-2026 Print
Safety is the most critical requirement in the development of battery technology. Ensuring no thermal runaway under thermal abuse is the primary prerequisite for their practical application. In recent years, non-flammable electrolytes, represented by organic phosphates, have been playing a key role in battery safety due to their inherent non-flammable characteristics, making them one of the hottest research topics for high-safe lithium/sodium-ion batteries. However, simply focusing on non-flammability of the electrolyte alone cannot guarantee that a battery will not undergo thermal runaway. The critical challenge lies in resolving side reactions at the cathode and anode interfaces at high temperatures, as well as the crosstalk between the cathode and anode.
Currently, most non-flammable electrolytes rely on high-concentration/localized high-concentration or fluorinated systems, resulting in high costs and poor compatibility with hard carbon anodes, which severely limits their practical application. Particularly for sodium-ion batteries, most reported studies have focused primarily on verifying the non-flammability of the electrolyte itself, lacking electrochemical and thermal safety validation in ampere-hour (Ah)-level cells. Therefore, developing a low-cost non-flammable electrolyte with excellent interfacial stability that can ensure superior electrochemical performance while achieving non thermal runaway in Ah-level cells remains one of the key challenges to be overcome in the development of high-safe sodium-ion batteries.
To address the aforementioned challenges, a team led by Prof. HU Yong-Sheng at Institute of Physics, Chinese Academy of Sciences, recently proposed a new polymerizable non-flammable electrolyte (Polynonflyte, or PNE). Based on a dual-salt system of sodium tetrafluoroborate (NaBF₄) and sodium hexafluorophosphate (NaPF₆), the team optimized the solvation structure and constructed a Boron (B)-containing cathode electrolyte interphase (CEI) and a PO₂⁻-rich solid electrolyte interphase (SEI) on the anode, successfully resolving the compatibility challenge with hard carbon anodes. While maintaining excellent electrochemical performance, the electrolyte capitalizes on the high-temperature endothermic decomposition and "thermal-induced self-polymerization" properties of the triethyl phosphate (TEP) solvent. This allows the electrolyte to be solidified rapidly during abnormal temperature rises in the battery, thereby blocking crosstalk between the cathode and anode and eliminating thermal runaway reactions.
Based on this strategy, the team conducted thermal safety validations in 3.5 Ah-level sodium-ion battery cells. At 100% state of charge (SOC), the cells successfully passed the nail penetration test without emitting smoke, catching fire, or exploding. In accelerating rate calorimetry (ARC) and thermal abuse tests, no thermal runaway occurred even at 300℃. Furthermore, the sodium-ion cells based on the PNE electrolyte exhibit excellent cycling stability, a wide operating temperature range (-40℃ to 60℃), and high-voltage stability (>4.3V), providing a vital foundation for the development of high-safe sodium-ion batteries.
This study was published at Nature Energy under the title "Thermal runaway-free ampere-hour-level Na-ion battery via polymerizable non-flammable electrolyte".
The study was supported by projects including the National Key R&D Program of China (2022YFB2402500), the National Natural Science Foundation of China (52394170-52394174, 22522310, and 22339001), Commanding heights of science and technology of Chinese Academy of Science (LDES150000), the Strategic Priority Research Program of the Chinese Academy of Sciences (XDA0400000), the Beijing-Tianjin-Hebei Basic Research Cooperation Project (B2024208091), the Jiangsu Provincial Carbon Peak and Neutrality Innovation Program (BE2022002-5), the International Partnership Program of the Chinese Academy of Sciences (005GJHZ2023021MI), and the 10th Young Elite Scientists Sponsorship Program by CAST.
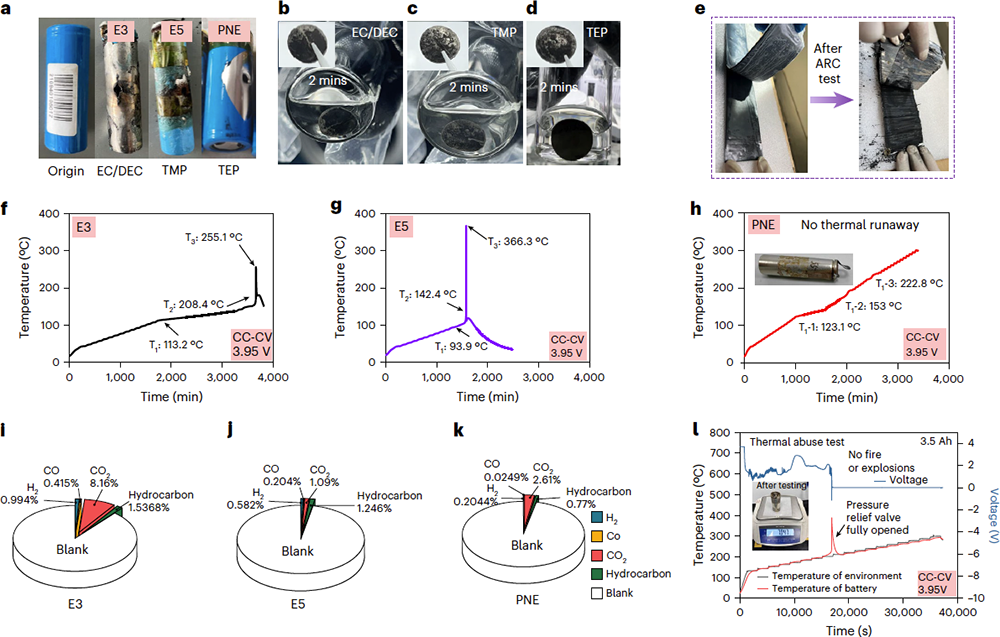
Fig.1 Safety tests of Ah-level sodium-ion batteries based on PNE. (Image by Institute of Physics)
Contact:
Institute of Physics
HU Yong-Sheng
Email:yshu@iphy.ac.cn
Key words:
Sodium-ion batteries; Safety; No thermal runaway;
Abstract:
The team led by HU Yong-Sheng at the Institute of Physics, Chinese Academy of Sciences has shattered the traditional perception that "non-flammability equals safety". Moving beyond a single line of defense, they constructed a "three-in-one" intelligent safety protection system encompassing "thermal stability, interface stability, and physical isolation". The team successfully developed a polymerizable non-flammable electrolyte (PNE) with self-protective functions, achieving a safety breakthrough of "no thermal runaway" in Ampere-hour-level sodium-ion batteries for the first time in the world.


