Carbon Monoxide and Neon Gas Encapsulated into H2O Cages: Kinetics and Dynamics Study of CO and Ne Clathrate Hydrate
Date:30-09-2014 Print
Clathrate hydrates have an ice-like crystalline structure formed by water in hydrogen-bonded frameworks and “guest” gas molecules in the framework cavities, usually under low-temperature and high-pressure conditions. The schematic picture of clathrate hydrate is shown in Fig.1 left.
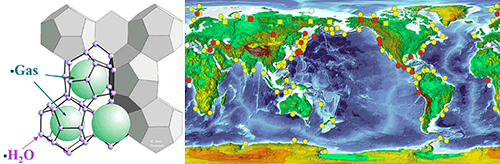 |
| Fig.1: the schematic picture of clathrate hydrate (left); the nature gas clathrate hydrate distribution on earth (right). |
Despite the importance of clathrate hydrate in energy and environmental sciences, some phase diagram properties of gas hydrates as well as their formation and decomposition kinetics are neither well-known nor properly understood. Recently, Dr. Xiaohui Yu, Dr. Jinlong Zhu, Prof. Changqing Jin and Prof. Yusheng Zhao, from Beijing National Laboratory for Condensed Matter Physics at the Institute of Physics, Chinese Academy of Sciences studied the CO and Ne gas hydrate with in-situ high pressure low temperature neutron diffraction.
Based on the time-dependent study of the CO clathrate hydrate formation in the CO-H2O system, they have demonstrated that sII hydrate can be formed in a time-evolving sequence after sI hydrate has initially crystallized, Fig. 2 left. This finding validates previous hypotheses that sII CO hydrate would become more stable than sI CO hydrate when the concentration of CO molecules is saturated. This behavior is associated with the difference in CO binding energy between 51262 and 51264 cages, where the 51264 cage in the sII structure is energetically favored over the 51262 cage in the sI structure for double occupancy of CO molecules. More importantly, this is attributed to the crossover in the binding energy-cage occupancy space between the two cage types. As a result, a sII hydrate enclosing two CO molecules in 51264 cages can be stabilized at certain P–T conditions through kinetically controlled cage filling, Fig. 2 right. However, the (CO)2-(H2O)28 clusters in an isolated state are energetically unfavorable and can readily dissociate into CO-(H2O)28 and CO. Our MD simulations suggest that the interactions between adjacent cages including CO-H2O and CO-CO interactions provide a significant source of stability for the double-CO occupancy of hexakaidecahedral cage. This work has been published in Nat. Comms. 5, 4128 (2014).
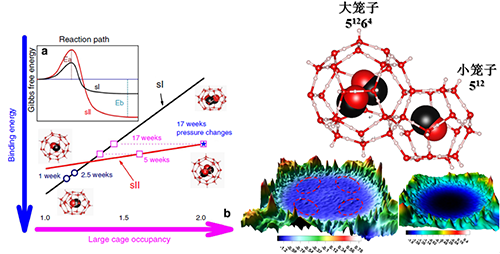 |
| Fig.2: CO clathrate hydrate formation kinetics (left); the sII structure CO Clathrate hydrate structure(right). |
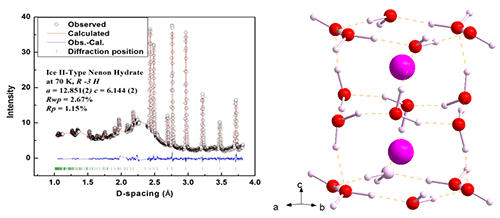 |
| Fig. 3: neutron diffraction pattern of Ne clathrate hydrate (left); the refined ice-II structure Ne hydrate (right). |
Contact information:
Dr. Xiaohui Yu
Email: yuxh@iphy.ac.cn
Dr. Jinlong Zhu
Email: jlzhu04@iphy.ac.cn
Prof. Yusheng Zhao
Email: yzhao77@iphy.ac.cn
Institute of Physics, Chinese Academy of Sciences


